A huge thank you to everyone that has contributed to our fabric mask material testing campaign! The results from the testing campaign are finally in. We tested two different 3-layer fabric combinations to establish baseline performance for fluid resistance, breathability, bacterial filtration efficiency (~3um particles), and submicron particle efficiency (~0.3um). These tests focused on two fabric combinations:
- 3-layers of spunbond nonwoven polypropylene (NWPP x 3)
- a 3-layer combination of cotton and spunbond NWPP (cotton-NWPP-cotton)
The tests were conducted by an accredited lab so we could see how the two different fabric mask material combinations measure up to both traditional medical mask material standards (ASTM F2100/EN1683) and the new barrier face covering standards (ASTM F3502).
Key Observations
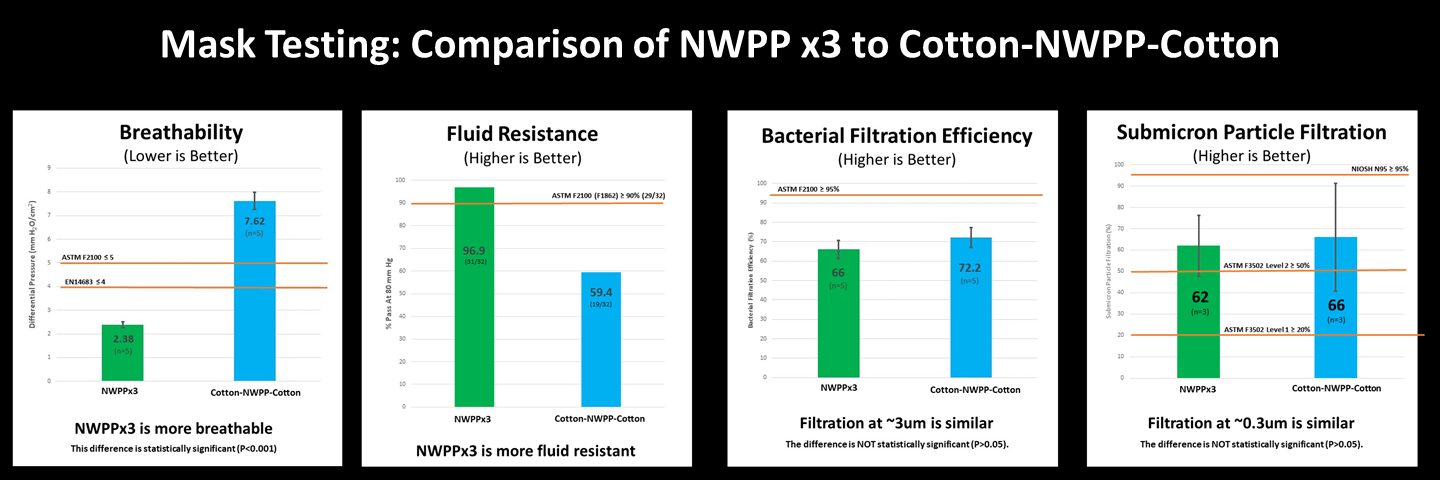
Overall, spunbond nonwoven polypropylene (NWPP) performed well. While the filtration of 3-layer NWPP samples was similar to a 3-layer Cotton-NWPP-Cotton samples, the breathability of the three-layer NWPP was 3 times better, and NWPP has the added benefit of being more fluid resistant. In addition, preliminary evidence suggests that mask performance with both material combinations is consistent with the new ASTM Barrier Face Covering standard for Level 1 breathability and filtration. Click on the image to enlarge, and read about the details below.
Table of Contents
- What was the overall goal of this testing?
- What was the specific objective of this round of testing?
- Which materials/combinations did we test?
- Part 1: Performance of samples relative to medical mask standards (ASTM 2100/EN14683)
- Part 2: Performance of masks relative to barrier face covering standards (ASTM F3502)
- Conclusion
What was the overall goal of this testing?
The overall goal of this testing was to provide open access, quantitative, head-to-head (i.e., apples-to-apples) data that is consistent with regulatory testing standards. The testing results can help guide fabric mask makers in selecting more effective materials and layering combinations for their masks for COVID-19.
Which materials/combinations did we test?
Test samples were constructed from either three layers of spunbond NWPP (204 gsm total) or a combination of spunbond NWPP and cotton (362 gsm total). The NWPP selected was Smart-fab® DoubleThick NWPP, 68 gsm, and the cotton was Robert Kauffman Kona® Natural, 147 gsm. See the table below for details:
| Design | Mask Layers | ||
| Skin Contacting | Middle | Outer | |
| NWPP x3 | NWPP (68 gsm) | NWPP (68 gsm) | NWPP (68 gsm) |
| Cotton-NWPP-Cotton | Cotton (147 gsm) | NWPP (68 gsm) | Cotton (147 gsm) |
For more information about how/why these materials were selected, please see “Finding and Selecting Polypropylene for Fabric Masks.”
Part 1: Performance relative to medical mask standards (ASTM 2100/EN14683)
The specific objective of Part 1 of the testing was to evaluate our selected material combinations for fluid resistance, breathability, and bacterial filtration in accordance with standards for commercial medical masks (e.g., surgical masks). For each test below, we summarize performance relative to both the American standard, ASTM F2100, and the European standard, EN14683, for medical masks:
- Fluid resistance — Do the samples block liquid splashes and sprays?
- Breathability — Do the samples allow air to freely pass through them?
- Bacterial filtration efficiency — Do the samples filter mid-sized droplets and particles (~3 um)?
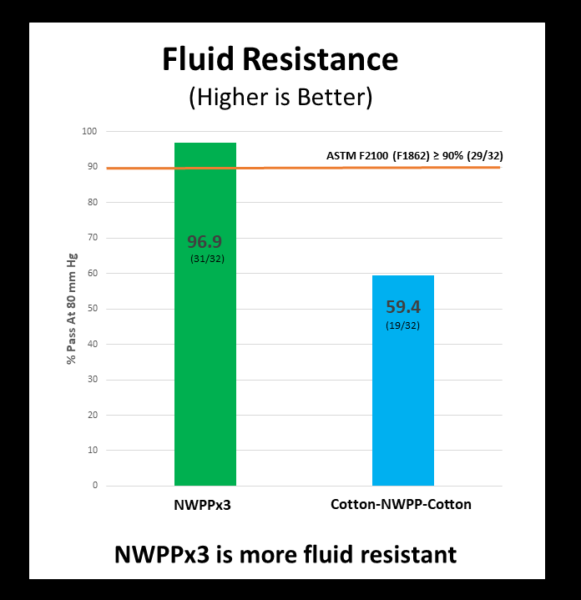
Fluid Resistance – Do samples block splashes and sprays?
When it comes to blocking splashes and sprays, the NWPPx3 performs significantly better than the Cotton-NWPP-Cotton.
This fluid resistance testing was done by an accredited lab (Nelson Laboratories-Fluid Resistance) in accordance with ASTM Level 1 standards. The ASTM Level 1 standard for fluid resistance, requires that at least 29 out of 32 samples (90%) of each fabric combination meet the standard. The NWPPx3 combination passed (31/32) while the Cotton-NWPP-Cotton did not (19/32). See the table below for details. Click the selected sample types for the formal laboratory report:
| Fluid Resistance @ 80 mm Hg | Lab Result (% Pass) | Medical Masks | |
| ASTM F1862 (Level 1: @ 80 mm Hg; ≥ 90%) | EN14683 (Type I: Not Required) | ||
| NWPP x3 | 94% (31/32) | PASS | N/A |
| Cotton-NWPP-Cotton | 59% (19/32) | FAIL | N/A |
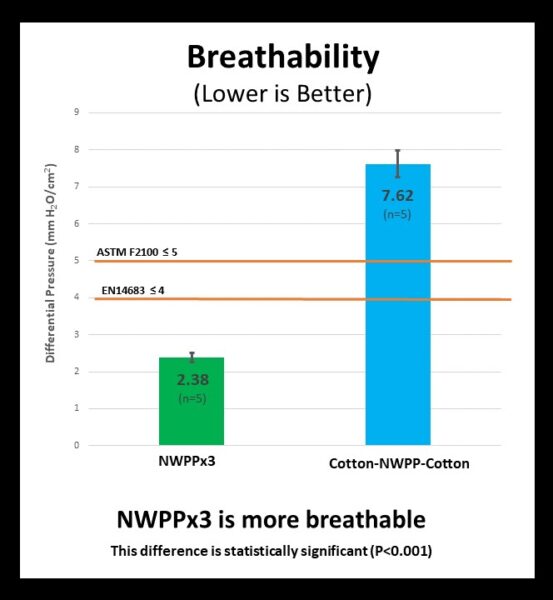
Breathability – Do the samples allow air to freely pass through them?
Medical mask standards for breathability are quite demanding and only the NWPPx3 combination met this high standard while the Cotton-NWPP-Cotton combination did not. This means that the NWPPx3 combination is considered easier to breathe through than the Cotton-NWPP-Cotton combination.
The breathability of five samples of each fabric combination was tested by an accredited lab (Nelson Laboratories). All five of the NWPP x3 masks satisfied the ASTM 2100 requirement for less 5 mm H2O/cm2 pressure difference, whereas none of the Cotton-NWPP-Cotton samples did. The difference in the breathability between the NWPPx3 and the Cotton-NWPP-Cotton is statistically significant (P< 0.05). See the table below for details, or click the selected sample type below for the formal laboratory report.
| Breathability (∆P) | Lab Result (mm H20/cm2) | Medical Masks | |
| ASTM F2100 (< 5 mm H20/cm2) | EN14683 (< 4 mm H20/cm2) | ||
| NWPP x 3 layers | 2.4 ± 0.1 (n=5) | PASS | PASS |
| Cotton-NWPP-Cotton | 7.6 ± 0.4 (n=5) | FAIL | FAIL |
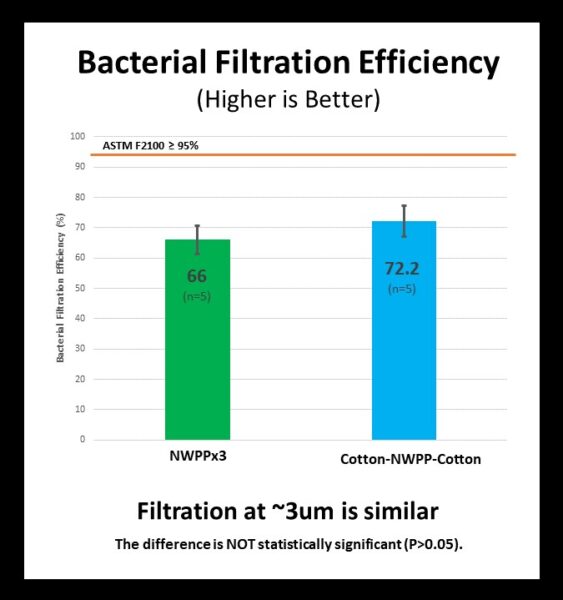
Bacterial Filtration Efficiency (BFE) – Do samples block mid-size droplets and particles (~3um)?
Although neither set of mask fabrics satisfy the bacterial filtration efficiency requirements for medical masks, both sets of materials perform similarly and filter out more than 50% of particles ~3um in diameter.
As discussed in our upcoming post about filtration, filtration standards and test methods vary widely and it is important to pay attention to particle size and type when evaluating filtration data. Here we evaluated filtration using bacterial filtration efficiency (BFE), which is the standard for medical masks that focuses on the filtration of bacterial particles with a mean size of ~3um.
BFE was tested for the two sets of test samples by an accredited lab (Nelson Labs: Bacterial Filtration Efficiency). For this data set, the mean performance of the Cotton-NWPP-Cotton was slightly better than that of the NWPPx3. However, this difference was not significant (P > 0.05). See the table below for details, or click the selected sample type below for the formal laboratory report.
| Bacterial Filtration Efficiency (BFE) | Lab Result (% Filtration) | Medical Mask | |
| ASTM F2100 (≥ 95%) | EN14683 (≥ 95%) | ||
| NWPPx 3 | 66 ± 5 (n=5) | FAIL | FAIL |
| Cotton-NWPP-Cotton | 72 ± 5 (n=5) | FAIL | FAIL |
Part 2: Performance relative to barrier face covering standard (ASTM F3502)
The specific objective of Part 2 of the testing was to evaluate mask prototypes constructed from the same material combinations as above for breathability and submicron particle filtration (~0.3 um), consistent with the new ASTM standards for barrier face coverings. (Read more about ASTM F3502 here.)
- Breathing resistance – Do the prototypes allow air to freely pass through them?
- Submicron particle filtration (PFE) – Do prototypes block small-size droplets and particles (~0.3um)?
It is important to note that to satisfy the requirements of the new standards, a total of ten samples of each fabric combination would need to be tested. For this testing, a representative sample of three masks constructed from each fabric type was evaluated.
Breathing Resistance – Do the masks allow air to freely pass through them?
All of the masks tested (both NWPP x3 and Cotton-NWPP-Cotton) satisfied the Level 1 breathability criteria for ASTM F3502. However, only the NWPP x3 masks satisfied the more stringent Level 2 breathability criteria. The difference in breathability between the NWPP x3 and the Cotton-NWPP-Cotton masks is statistically significant (p<0.05). Consistent with our results above, this means that the NWPP x3 masks are considered easier to breathe through than the Cotton-NWPP-Cotton masks, and are likely to be more comfortable for long duration wear and/or use during high intensity activities.
See the table below for details, or click the selected sample type below for the formal laboratory report.
| Breathing Resistance | Lab Result (mm H2O) | ASTM F3502 Barrier Face Coverings | ||
| Mean | Maximum |
Level 1 Maximum ≤ 15 mm H20/cm2 |
Level 2 Maximum < 5 mm H20/cm2 |
|
| NWPP x 3 layers | 1.7 ± 0.2 (n=3) | 1.9 | PASS | PASS |
| Cotton-NWPP-Cotton | 6.3 ± 0.5 (n=3) | 6.8 | PASS | FAIL |
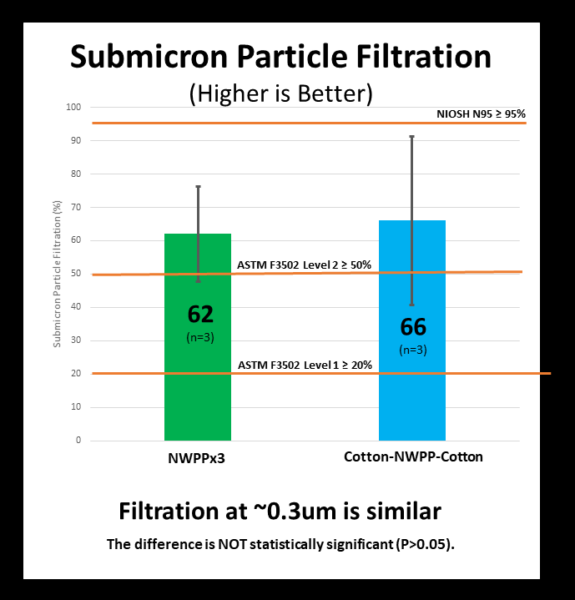
Submicron particle filtration (PFE) – Do masks block small-size droplets and particles (~0.3um)?
All of the masks tested (both NWPP x3 and Cotton-NWPP-Cotton) satisfied the minimum performance criteria for Level 1 (≥ 20%) submicron filtration according to ASTM F3502. In fact, all of the masks tested (6/6) had submicron filtration more than double the minimum required filtration (>40%) for ASTM F3502. This means that both fabric combinations performed reasonable well on submicron particle filtration.
Although most of the masks tested exceeded the higher performance Level 2 (≥ 50%) filtration criteria, the ASTM 3502 standard requires that the value from the worst performing mask of each set be reported. As a result, when reporting performance relative to the standard, neither fabric combination satisfied the Level 2 performance requirement since the lowest performance for the NWPP x3 was 46.2% and for the Cotton-NWPP-Cotton it was 41.7%.
| Submicron Filtration | Mask | Lab Result | ASTM F3502 Barrier Face Coverings | |
| Level 1
≥ 20% |
Level 2 ≥ 50% |
|||
| NWPP x3 | 1 | 46.2 | PASS | FAIL |
| 2 | 66.1 | PASS | PASS | |
| 3 | 73.8 | PASS | PASS | |
| Cotton-NWPP-Cotton | 1 | 64.3 | PASS | PASS |
| 2 | 92.1 | PASS | PASS | |
| 3 | 41.7 | PASS | FAIL | |
There was a wide range in performance within each type of mask evaluated as evidenced by the table above and the size of the standard deviations (table and graph below). Based on conversations with the lab, the lower performing masks did not seal as well to the testing apparatus. As such, the minimum performance reported may be an underestimate of real-world performance. This may not be surprising given masks were designed to fit to the contours of the face and not a round opening on a flat surface (e.g., the testing apparatus). However, this result serves as a reminder of the importance of mask fit, and not just the performance of mask fabrics evaluated in isolation.
See the table below for details, or click the selected sample type below for the formal laboratory report.
| Submicron Filtration | Lab Result | ASTM F3502 Barrier Face Coverings | ||
| Mean | Minimum | Level 1
Minimum ≥ 20% |
Level 2 Minimum ≥ 50% |
|
| NWPP x3 | 62 ± 14 % (n=3) | 46.2% | PASS | FAIL |
| Cotton-NWPP-Cotton | 66 ± 25 % (n=3) | 41.7% | PASS | FAIL |
Conclusion:
In this post we compared two mask fabric combinations, NWPP x3 (204 gsm total) and Cotton-NWPP-Cotton (362 gsm total), to medical mask standards for fluid resistance, breathability, and bacterial filtration efficiency, as well as barrier face covering standards for breathability an submicron particle filtration.
- Relative to medical mask standards: We found that NWPP x3 performed better than Cotton-NWPP-Cotton in terms of breathability and fluid resistance, and that both combinations performed similarly on filtration. It is important to note that neither combination satisfied all the requirements for use in high-risk medical situations.
- Relative to barrier face covering standards: All of the masks tested from both combinations (NWPP x3 and Cotton-NWPP-Cotton) pass the minimum criteria for the new ASTM F3502 standards for barrier face coverings in terms of both filtration and breathability. As with the testing relative to medical mask standards, the NWPP x3 combination significantly outperformed the Cotton-NWPP-Cotton on breathability, and both combinations performed similarly on filtration.
Final Take-Aways:
These results provide valuable information for community mask users and mask makers. Our take-aways:
- For high-intensity activities (e.g., athletic activities) and longer duration activities, masks constructed with more breathable NWPP layers and fewer cotton layers may be more comfortable.
- For lower intensity and/or shorter duration activities (e.g., shopping), masks made from either combination should be appropriate.
- For non-medical situations where you may be exposed to splashes or sprays (e.g., sneezes, or toddlers still learning to “say it, don’t spray it”), the NWPPx3 combination may be a better choice.
| Standard | Test | NWPP x3 | Cotton-NWPP-Cotton | |
| Medical Mask (EN14683/ASTM 2100) | Fluid Resistance | 80 mm Hg | PASS | FAIL |
| Bacterial Filtration Efficiency | ≥ 95% | FAIL | FAIL | |
| Breathability | < 5 mm H20/cm2 | PASS | FAIL | |
|
Barrier Face Covering (ASTM F3502) |
Breathability | Level 1 (Adequate) | PASS | PASS |
| Level 2 (Excellent) | PASS | FAIL | ||
|
Submicron Particle Filtration |
Level 1 (Adequate) | PASS | PASS | |
| Level 2 (Excellent) | FAIL | FAIL | ||
Thanks again to everyone that contributed to our mask testing campaign!





As a seamstress, I wouldn’t use Kaufman Kona for a mask. As its weight should indicate, it’s very heavy and thick, unsuitable for a face covering. It makes nice, thick, opaque blouses and skirts and even trousers. It absorbs water readily and makes nice tea towels and napkins. The masks made for testing must have been unwearable, with big lumpy seams where multiple layers overlapped. It’s woven from a slightly fluffier thread than other cottons with similar TPI.
A lighter weight but equally high or higher TPI (threadcount) (thin, but tightly woven) cotton would have been a significantly better choice. Also using more than one existing standard of textile terminology (weight only describes one thing, and not a very useful one for this purpose) would be very helpful. Some examples of other fabrics (with TPI info as well as weight) can be found here https://www.dharmatrading.com/fabric/finely-woven-fabrics.html?lnav=fabric_cotton.html